1) The role of Schwann cells in pathologic scar formation
The skin is one of the largest and most versatile organ in the human body. It performs metabolic functions, participates in immune defense, and protects the body from pathogenic factors. Whenever there is an injury to the skin, the body initiates an immediate healing process, which involves numerous cell types and factors that aid in proper wound healing and scar formation. However, scar formation is often hampered, leading to the development of hypertrophic or keloid scars. The reasons for such differences in scar formation are largely unknown.
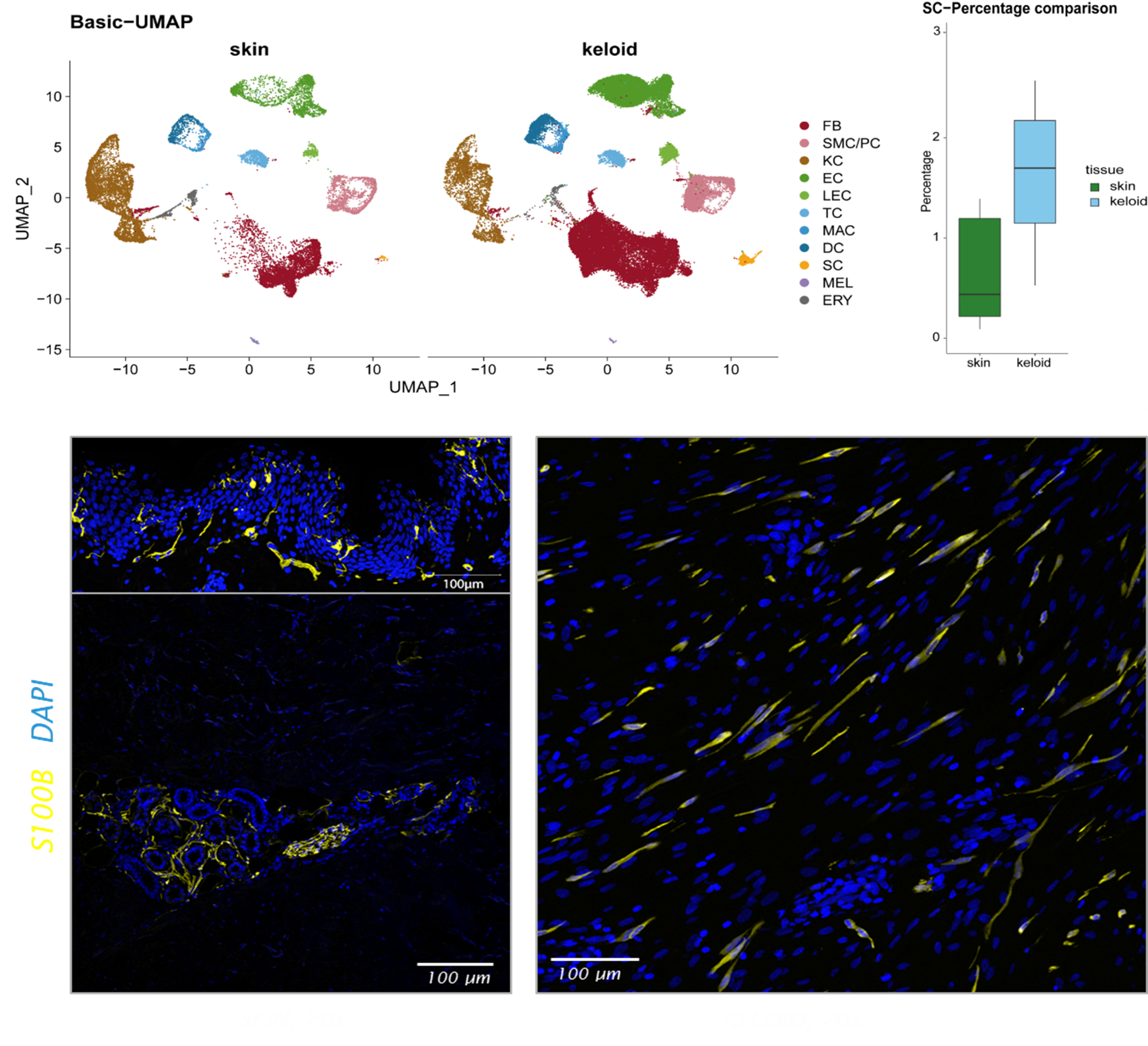
Based on our previous research, we have found that Schwann cells, which are cells of the nervous system that surround axons, play a critical role in the development of abnormal scars. In these scars, Schwann cells lose contact with axons and instead interact with M2 macrophages and chondrocytes to maintain their de-differentiated and repair-like phenotype. Thereby, they contribute significantly to the progression of pathologic scars by massive production of the extracellular matrix.
2) Development of novel 3D in vitro models of hypertrophic/keloid scars
The primary challenge in scar research is the absence of adequate models that accurately mimic human pathological scars in both in vitro and in vivo settings. Thus, our primary objective is to establish an in vitro model that closely replicates the situation observed in human pathological scars. Additionally, we aim to comprehensively analyze the interaction between Schwann cells, macrophages, chondrocytes and fibroblasts, and to identify novel factors involved in the pathogenesis of hypertrophic and keloid scar formation. Understanding these factors could provide new insights for the development of novel therapeutic interventions.
3) Secretome-based wound healing
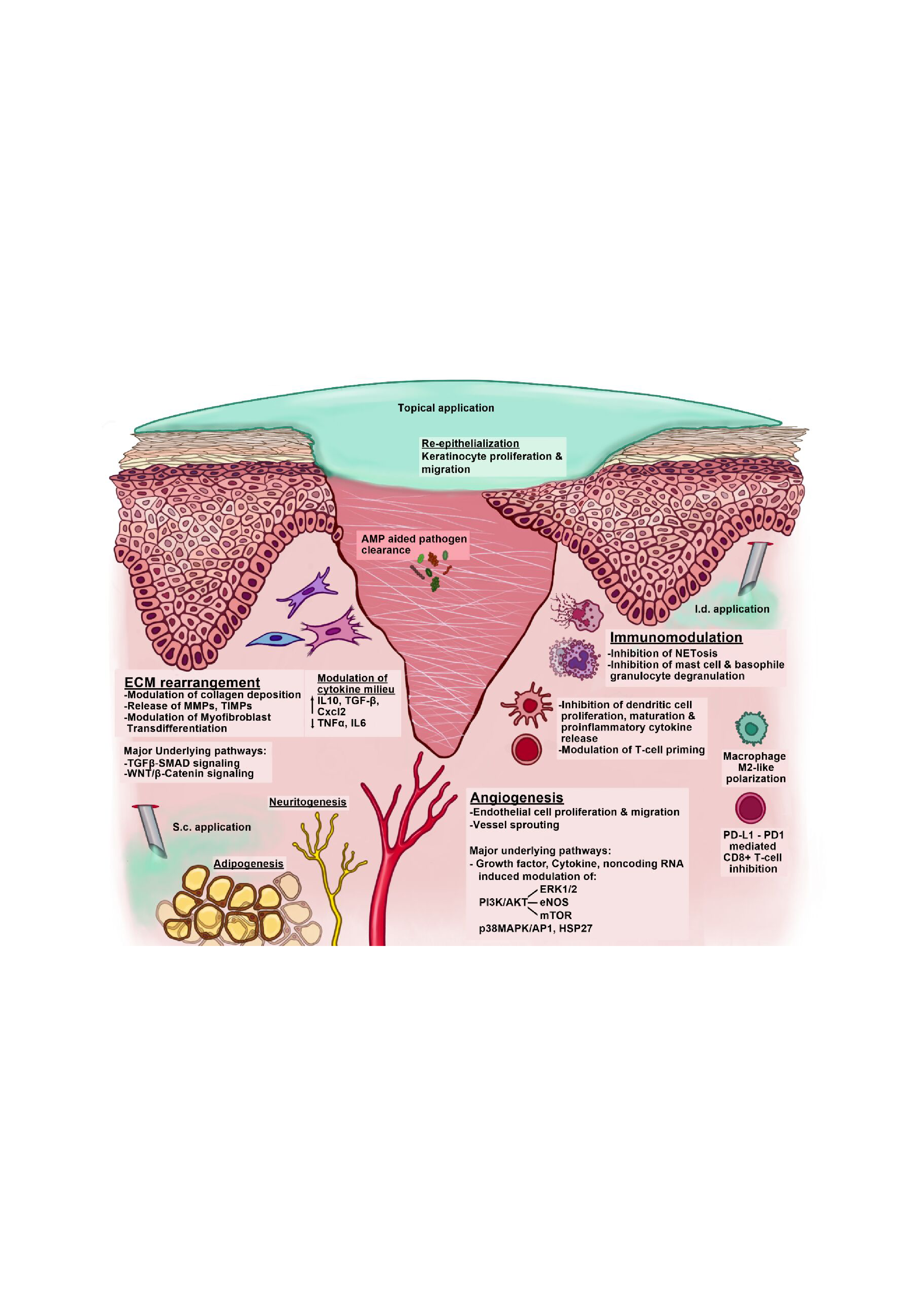
Although application of stem cells to chronic wounds emerged as a candidate therapy in the previous century, the mechanism of action remains unclear. Recent evidence has implicated secreted paracrine factors in the regenerative properties of cell-based therapies. In the last two decades, considerable research advances involving the therapeutic potential of stem cell secretomes have expanded the scope of secretome-based therapies beyond stem cell populations.